Children with liver transplants must take immunosuppressant medications for life. To maintain tolerance of the transplanted liver, doctors treat transplant rejection and conduct ongoing maintenance immunosuppression by increasing dosages of medication enough to counteract rejection.
Children’s Hospital Los Angeles researchers are developing an imaging panel that identifies the cell types implicated in transplant rejection. Their goal is to find potential biomarkers that can lead to more personalized immunosuppression therapies that can treat rejection with fewer side effects. The researchers’ findings were published in Science Advances.
Current immunosuppressant medications are less than optimal, because they don’t specifically target the subpopulations of immune cells associated with T–cell-mediated rejection in liver transplant. Up to 10% of liver transplant patients develop steroid resistance and experience recurrent episodes of T–cell-mediated rejection. These drugs have toxicities and can cause severe side effects.
“We have not changed the way we treat rejection in 40 years,” says Juliet Emamaullee, MD, Ph.D., FRCSC, FACS, transplant surgeon at Children’s Hospital Los Angeles.
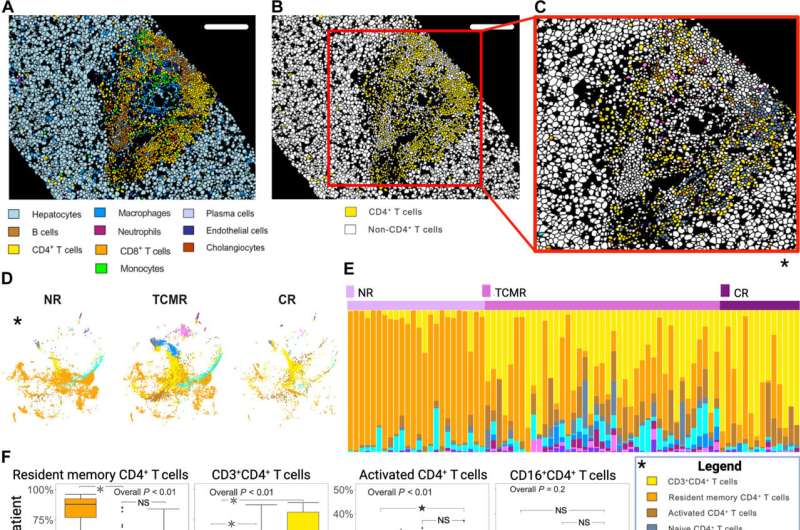
A multicenter team of researchers led by Dr. Emamaullee developed a proteomics-based imaging mass cytometry marker panel to categorize different immune cell types at single cell resolution. (The panel is multiplexed, meaning it can detect multiple biomarkers simultaneously.) They used tissue biopsies from 79 adult liver transplant patients—including patients who received transplants at CHLA before age 18—who had either no rejection, acute T–cell-mediated rejection or chronic rejection.
From this data, the researchers generated an atlas of over 400,000 cells—comprising 42 different cell types. Using machine learning to characterize the types of immune cells interacting in the microenvironment during active T-cell-mediated rejection episodes, the researchers found that regulatory (HLA-DR+ Treg) and PD1+ T-cell phenotypes (CD4+ and CD8+ 50 subsets), combined with variations in M2 macrophage polarization, were unique signatures of active T–cell-mediated rejection.
“Inflammation in liver transplant rejection is very complex and there are certain rare populations of T cells that are unique to different phases of rejection,” says Dr. Emamaullee, Research Director, Division of Abdominal Organ Transplantation and study corresponding author.
“We repeatedly saw the same patterns of cell types involved in rejection and their spatial relationships. And now that we understand that those cells are causing injury in the tissue, it gives us an idea of how to find therapies that might target them and biomarkers that may reflect that cell population in the blood. Hopefully we can get away from biopsies altogether in our pediatric patients—we can just detect that those cells are in the liver by doing a blood test.”
More information:
Arianna Barbetta et al, Spatially resolved immune exhaustion within the alloreactive microenvironment predicts liver transplant rejection, Science Advances (2024). DOI: 10.1126/sciadv.adm8841
Citation:
Researchers probe immune microenvironment to prevent pediatric liver transplant rejection (2024, April 17)
probe-immune-microenvironment-pediatric-liver.html
.
. The content is provided for information purposes only.
